Frontiers What’s next for GLPs? Salk scientists are shaping the future of weight-loss medicine
Every day, our bodies are tasked with finding food, turning that food into usable energy, and then spending that energy wisely on things like breathing, moving, thinking, and healing. This series of processes, collectively known as metabolism, is coordinated by a complex network of genes. Switching on and off like an array of microscopic levers, these genes tell our cells when to store energy, when to burn it off, and whether to tap into fat, muscle, or sugars for fuel.
This exquisitely balanced system worked well when humans spent their lives farming fields, hunting for food, and traveling on foot, but in the modern world, it seems to be breaking down, says Professor Ronald Evans, holder of the March of Dimes Chair in Molecular and Developmental Biology at Salk.
“We’re seeing a global increase in weight and excess body fat, beginning at younger and younger ages,” he says. “We’re not sure exactly why it’s been happening, but it’s definitely not going backwards.”
Enter glucagon-like peptide-1, or GLP-1, a hormone that is produced naturally in the body and plays an important role in appetite suppression. But while GLP-1 agonists like Ozempic® and Wegovy® have exploded in popularity in recent years, Salk researchers, along with other scientists, have been studying GLPs for decades.
It’s long been known that GLP-1 is involved in regulating metabolism to control functions like calorie intake and sugar levels. Now, Evans and other Salk faculty are using their research on GLPs and the basic science of metabolism to help advance the next generation of GLP-based weight-loss therapies.
“What we’re starting to see is that every nutrient we consume takes its own path through the body, and any drug we take or any disease we have tends to alter that,” says Professor Christian Metallo, holder of the Daniel and Martina Lewis Chair at Salk. “The more we know about this system at the molecular level, the better we can understand and control it.”
Room to Improve on GLPs
For many patients struggling with obesity, GLP-1 medications have been transformative. By treating their metabolism, these drugs empower sustainable long-term weight loss and reduce the risk of heart disease, diabetes, cancer, and a host of other dire long-term outcomes.

But the drugs are far from perfect.
“It is a blockbuster drug for diabetes and obesity, but we are well aware of its side effects,” Evans says.
Along with issues like nausea and vomiting—a common complaint that causes many patients to stop the medication and regain the weight—muscle loss is another serious concern.
In fact, about 40 percent of the weight people typically lose on GLP-1 drugs comes from lean mass, primarily muscle tissue. This can lead to weakness and increased risk of frailty, especially in older adults. And while doctors typically recommend exercise to people taking these drugs, muscle loss can actually make some exercises more challenging or less effective.
Other, less well-understood side effects of GLP-1 drugs include reports of retinal changes and vision problems, as well as mental and cognitive changes. Metallo says he expects more side effects to emerge as more people take GLP-1 agonists and new versions of the drugs enter the market.
“As we’re working on the next generation of these drugs, the big question is how we can stave off their potential side effects,” he says.
“Meal In a Pill”
GLP-1 is normally released in response to food; the hormone sends a signal that the body has had enough and tells the brain to stop eating. The current generation of weight loss drugs works by injecting a synthetic version of GLP-1 into the body to suppress appetite. Evans’ lab is working on a receptor called FXR that stimulates the body to produce GLP-1 naturally.
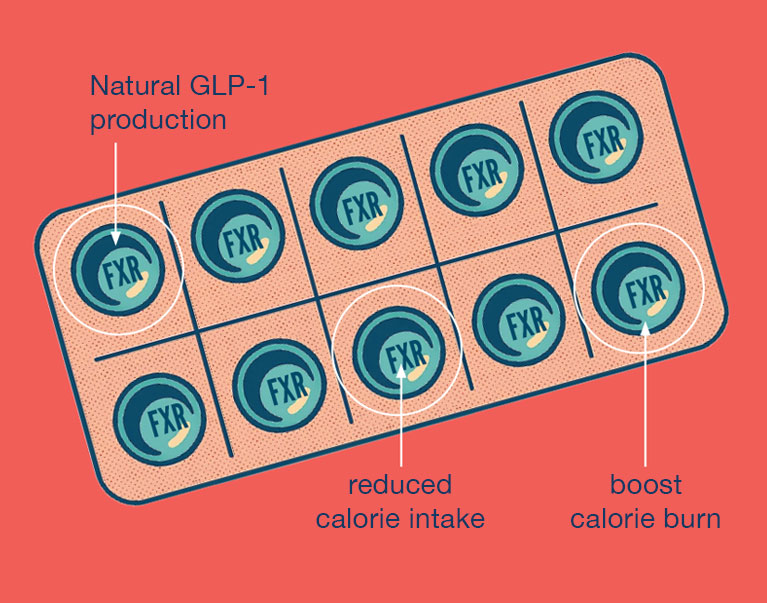
“We call these FXR compounds our ‘Meal in a Pill,’” Evans says, “because they activate genes that would normally be activated during a meal, but there is no meal—we’re activating it with a compound.”
This approach potentially offers several advantages over the current class of synthetic GLP-1 drugs.
“It’s not simply mimicking GLP-1,” says Weiwei Fan, a staff scientist in Evans’ lab. “It’s triggering a more natural response.”
Natural GLP-1 molecules break down quickly, but synthetic versions have a much longer half-life. This prolonged exposure enhances appetite suppression, but it may also cause many of the unwanted side effects.
In contrast, FXR drugs stimulate natural GLP-1 production in the body. The GLP-1 produced this way would be broken down faster, just like it would after a meal. This would deliver all the appetite-suppressing benefits without keeping the hormone in your system for so long that it starts to trigger side effects. What’s more, patients could take FXR drugs as a daily pill instead of the injections required for synthetic GLPs.
Activating the FXR pathway also fights obesity in two different ways. In addition to suppressing appetite through GLP-1 production, it also releases a second hormone, called fibroblast growth factor-19, that directs the body to burn energy.
“It’s cutting down calorie intake, and at the same time it’s boosting the burning of calories in the body,” Fan explains. “It’s targeting the obesity problem from both sides of the energy balance.”
Researchers hope this could help people lose weight with less muscle loss, Fan says. He notes that, in mouse studies of FXR, most weight loss came from fat, and lean muscle mass was not significantly affected.
“We think this is partly because FXR works by both reducing food intake and increasing energy expenditure,” Fan explained. “That means you won’t need as much reduction in food intake to reach the same weight loss goal, because a lot of this boosted energy expenditure is upon fat.”
In clinical trials, FXR is already showing promise as a target for fatty liver disease, another serious metabolic disorder, and Salk researchers are in discussions with pharmaceutical companies about developing an FXR-based obesity drug.

Preventing Muscle Loss
Salk researchers are also tackling the issue of muscle loss. Metallo notes that the way GLP-1 drugs suppress appetite leads the body to enter a kind of starvation state.
“Your body responds not just by burning fat; it actually starts to break down muscle and other proteins,” he says. “What needs to be better understood is, when you go through that prolonged starvation state, what else is being burned beyond the fat?”
It’s also easier for the body to make more fat than it is to make muscle, which is one reason why people who go through yo-yo diets can find it progressively harder and harder to keep the weight off, he notes.
Metallo’s lab studies how amino acids, the building blocks of proteins, are also used to make important fatty compounds called sphingolipids. He has zeroed in on the amino acids serine and glycine, which play an important role in supporting muscle regeneration, retinal health, and the peripheral nervous system.
The team’s research found that serine and glycine are broken down by an enzyme produced in the liver, and they’re now working on approaches to target that enzyme. The goal is to develop a drug that could be given alongside GLPs to help prevent muscle loss as people lose weight.
“Ideally, you want to trigger the body to maintain its energy levels by burning fat, instead of amino acids—especially these amino acids, which we know are critical in both muscle tissue and the nervous system,” Metallo says.
Investigating Other Pathways
Evans’ team is also interested in another receptor called PPAR-delta, which helps stimulate genes involved in fat metabolism. Activating this receptor causes the body to expend energy (burn calories) not through movement but by generating heat.
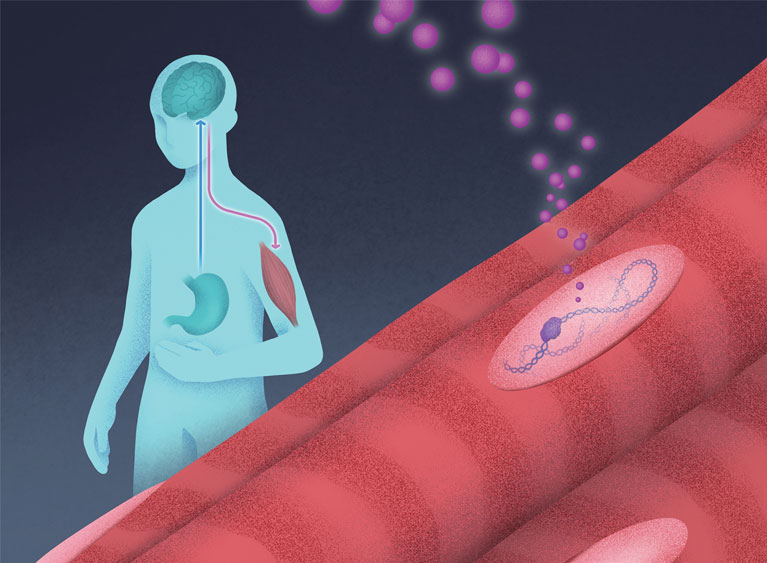
When the body is hungry, the brain sends growth hormone (pink) to protect the muscle tissue. This allows BCL6 (purple) to bind DNA and regulate genes for muscle preservation. Researchers are now exploring BCL6’s role in maintaining muscle during weight loss.
The researchers are also exploring a hormone receptor partner called BCL6, which plays a role in regulating muscle development and function.
“Our newest discovery shows that BCL6 is very important for maintaining muscle mass,” Evans explains.
Researchers are now investigating whether activating the BCL6 pathway could help prevent muscle loss in patients taking GLP-1 drugs.
“We’re very excited about these new targets, and we think they are opening up an entirely new opportunity in weight management,” he says. “It’s happening quickly, and I think it’s going to have a big impact.”
Salk scientists are also exploring how the fat in our adipose tissue is metabolized. They have found that certain fatty acids in the bloodstream correlate with insulin sensitivity, while others are linked to insulin resistance. An open question is whether dramatic changes to a person’s fat mass could affect that balance, Metallo says.
Looking to the Future of GLPs
For Salk researchers, the emergence of drugs like Ozempic and Wegovy doesn’t represent the culmination of GLP research—it marks the beginning.
“Everything we’re learning from the clinical use of GLP-1 drugs just motivates us even more to understand how every tissue and cell in our body handles nutrients, and how that changes in the context of disease,” Metallo says.
They also underscore the importance of basic science research to lay the foundation for new treatments that improve health and save lives. Evans pointed to his team’s work on FXR, PPAR-delta, and BCL6 as an example of where weight loss medicine could go next.
“These are pathways that literally were not known to exist until we discovered them,” he says. “This is how basic research really unlocks the future. Because we have advanced tools, we can find something new, quickly deconstruct what it is acting on, and then create modifications of that pathway to promote healthier function.”
Implications Beyond Obesity
Obesity research at Salk could also help advance treatments for other diseases.
The PPAR-delta pathway is an attractive target for obesity treatment because of its role in regulating energy expenditure, but there’s growing evidence that it is also important to other cell types, including microglia, a type of immune cell found in the brain.
“We think activating PPAR-delta in those brain immune cells may have major benefits for neurodegenerative diseases, including Alzheimer’s,” Fan says.
Evans’ team also recently completed a clinical trial in patients with cardiovascular disease, extending their track record of successful bench-to-bedside projects.
While stimulating certain metabolic pathways is being tested to promote muscle growth, blocking those same pathways could also slow cancer, Metallo says.
“I’ve learned a lot by jumping from disease to disease,” he says, “because in the end, the metabolism is the same—it’s just wired a little bit differently.”

Featured Stories
 Michelle Chamberlain: Bringing together communities old and newChamberlain, whose lifelong commitment to others has shaped her journey to Salk, now serves as Salk’s vice president of External Relations, leading fundraising, communications, community engagement, foundation relations, and stewardship efforts.
Michelle Chamberlain: Bringing together communities old and newChamberlain, whose lifelong commitment to others has shaped her journey to Salk, now serves as Salk’s vice president of External Relations, leading fundraising, communications, community engagement, foundation relations, and stewardship efforts. Joseph Swift: Saving potatoes, one road trip at a timeSwift, a plant biologist and startup co-founder, had an adventurous upbringing in Australia filled with natural beauty. Today, he uses plant genomics to tackle urgent questions in sustainability and agriculture.
Joseph Swift: Saving potatoes, one road trip at a timeSwift, a plant biologist and startup co-founder, had an adventurous upbringing in Australia filled with natural beauty. Today, he uses plant genomics to tackle urgent questions in sustainability and agriculture. Building connections in cells, soils, and labsAssistant Professors Lena Mueller and Aga Kendrick joined the Salk Institute in January 2024 and November 2023. Less than two years later, their new labs are already celebrating their first published studies.
Building connections in cells, soils, and labsAssistant Professors Lena Mueller and Aga Kendrick joined the Salk Institute in January 2024 and November 2023. Less than two years later, their new labs are already celebrating their first published studies. Empowering future scientists and advancing healthy aging researchThe Prebys Foundation empowers future scientists, while La Mer supports a fellowship advancing healthy aging research.
Empowering future scientists and advancing healthy aging researchThe Prebys Foundation empowers future scientists, while La Mer supports a fellowship advancing healthy aging research. Community building with creativity and connections across SalkIn April, Salk’s Engagement & Wellbeing Week brought employees and trainees together for activities ranging from coffee hours to a paper airplane competition.
Community building with creativity and connections across SalkIn April, Salk’s Engagement & Wellbeing Week brought employees and trainees together for activities ranging from coffee hours to a paper airplane competition.






















































